Citizen Request for Congressional Oversight
Psychedelic Research, Advocacy, and Commercialization Ecosystem
Medical standards of care do not happen in a vacuum—they begin with the systems that determine whose health is prioritized, whose pain is dismissed, and which research questions are funded. Based on publicly available reporting, concerns have been raised that psychedelic research may be constrained by institutional funding structures and governance overlap.
In recent years, calls for new psychedelic policy and expanded access have increased. At the same time, investigative reporting has highlighted questions about financial alignment, governance interdependence, and accountability within parts of the psychedelic research, advocacy, and commercialization ecosystem. Observing these dynamics concurrently has raised questions that may require independent oversight.
Call to Action for Congressional Submissions
In an effort to clear up any confusion, and determine the facts surrounding psychedelic science and commercialization, the letter in the next section has been submitted to my Congressional Rep requesting Congressional oversight.
As a Veteran, I urge you to do the same if these issues are of concern to you. It is disheartening to me when I hear that some folks feel as if there is nothing they can do, like they have no voice and no power. The irony in that position is that Veterans were told we were fighting for democracy, meaning the people have voice and power.
CONGRESSIONAL TEMPLATE: The issues listed in my example below focus on business activities that I believe to be a root cause. That said, there are many perspectives and potential issues. If you agree with my submission, please feel free to copy and paste to your respective rep. If you have additional or different perspectives, please consider writing your own version to submit.
FIND YOUR REP: https://www.house.gov/representatives/find-your-representative
We need eyes on from multiple perspectives and multiple districts. We need Congressional reps to start talking in the hallways and to start to compare notes. To ensure actual change, a single congressional may not be effective.
Citizen Request for Congressional Oversight
Psychedelic Research, Advocacy, and Commercialization Ecosystem
Submitted to: Committee Staff, U.S. House of Representatives
Purpose: Request for Oversight Review and Inquiry
Introduction
I respectfully submit this memorandum as a citizen request for congressional oversight, consistent with Congress’s constitutional responsibility to oversee federally regulated research, tax-exempt entities, and public health policy.
Summary of Concern
Investigative reporting and publicly available records indicate that:
Tax-exempt organizations, universities, and private foundations are closely interconnected with for-profit drug developers and investment vehicles
Financial ties between numerous organizations involved in the emerging psychedelic medicine ecosystem have been documented
Individuals appear to occupy overlapping roles as donors, board members, advisors, investors, and advocates
Research, training, credentialing, advocacy, and media activities may be coordinated with commercial pipelines approaching FDA approval
While collaboration across sectors is not inherently improper, the scale, concentration, and interdependence of these relationships raise legitimate public-interest questions that warrant congressional review. Taken together, these dynamics present questions in regard to regulatory exposure under tax laws and state charity laws, and create potential risks related to: 1) Improper private benefit from charitable assets, 2) Undisclosed or inadequately managed conflicts of interest, 3) Erosion of research independence, 4) Anticompetitive market structures, and 5) Public trust in federally regulated clinical research and healthcare policy.
These risks may not be apparent when reviewing individual entities in isolation, but become visible when the ecosystem is examined as a whole.
Why Congressional Oversight Is Requested
I respectfully request congressional attention for the following reasons:
Public interest and taxpayer impact: Significant federal tax benefits, charitable deductions, and public trust are involved
Novel industry structure: Psychedelic medicine uniquely blends philanthropy, academia, venture capital, advocacy, and media, often within the same governance networks
Regulatory precedent: Oversight outcomes may influence future FDA approvals, healthcare delivery models, and investment practices
Systemic risk: Potential compliance issues may emerge only at the network level, not through single-entity enforcement
International dimensions: Documented ties extend into Canada, raising cross-border oversight considerations
Requested Scope of Review
I respectfully ask Congress to consider whether a fact-finding inquiry is appropriate to assess:
Safeguards protecting the independence of clinical research
Compliance with federal and state nonprofit law
Integrity of tax-exempt research and educational activities
Adequacy of financial, governance, and conflict-of-interest disclosures
Use of charitable and public-interest resources
This request is intended to be preventative and informational, not accusatory.
Entities in Scope (Non-Exhaustive)
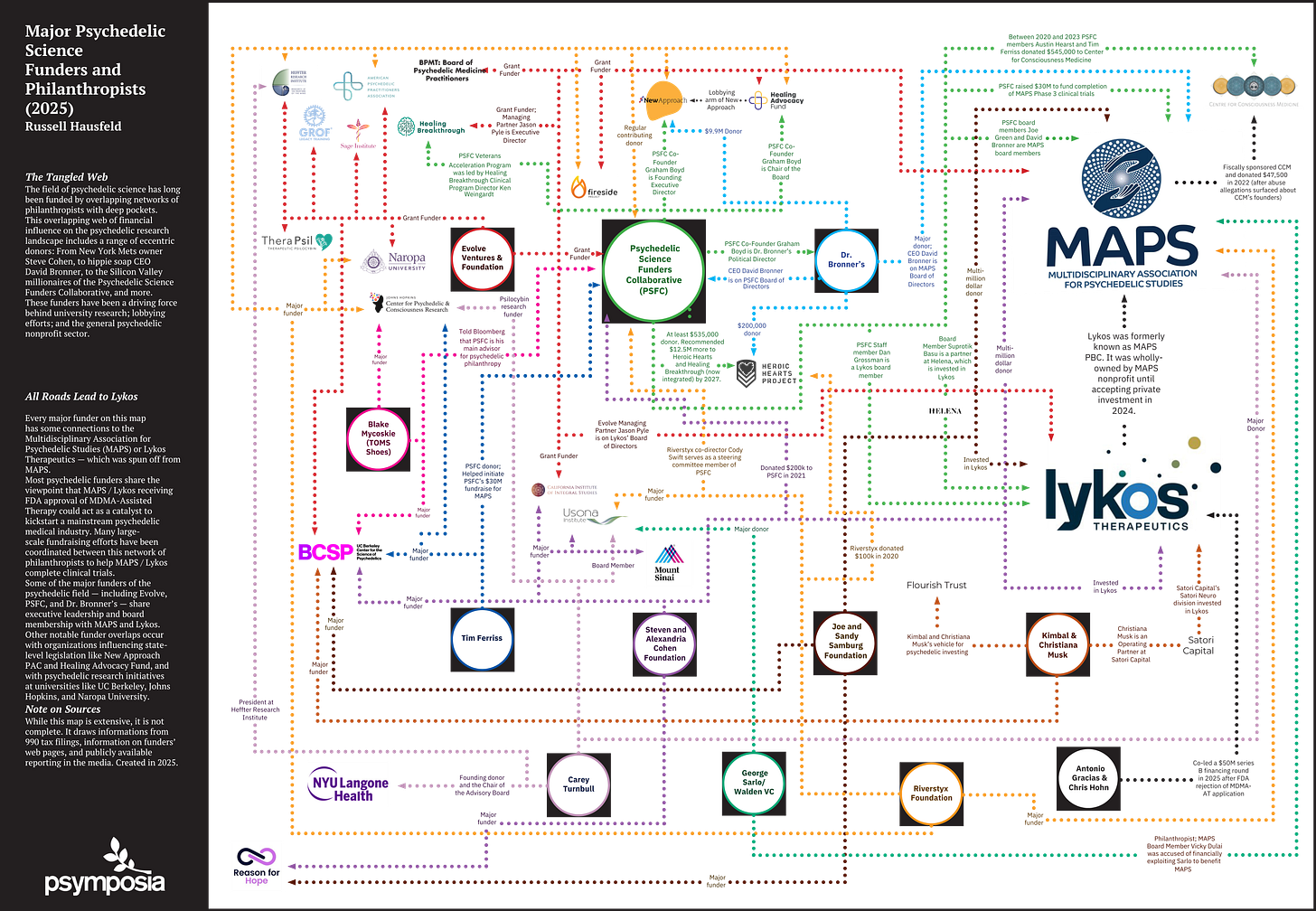
The following organizations and individuals are listed based on their appearance in the Psychedelic Funders Mind Map (2025), compiled by Psymposia using publicly available tax filings, organizational disclosures, and investigative reporting. Inclusion is for contextual transparency only and does not imply wrongdoing, coordination, or regulatory non-compliance.
Advocacy and Political Organizations
New Approach PAC (US); Healing Advocacy Fund (US)
Private Foundations and Trusts
Steven & Alexandra Cohen Foundation (US); Joe & Sandy Sandburg Foundation (US); Flourish Trust (US); Reason for Hope (US); Evolve Ventures & Foundation (US)
Venture, Corporate, and Pharmaceutical Entities
Resilient Pharmaceuticals (formerly Lykos) (US); Satori Capital (US); Helena Special Investments (US); Walden VC (US)
Nonprofit and Service Organizations
Multidisciplinary Association for Psychedelic Studies (MAPS) (US); Psychedelic Science Funders Collaborative (US); Heroic Hearts Project (US); RiverStyx Foundation (US); Fireside Project (US); TheraPsil (CA)
Academic and Research Institutions
Johns Hopkins Center for Psychedelic & Consciousness Research (US); UC Berkeley Center for the Science of Psychedelics (US); NYU Langone Health (US); Mount Sinai (US); Naropa University (US); California Institute of Integral Studies (US)
Training and Professional Organizations
American Psychedelic Practitioners Association (US); Board of Psychedelic Medicine Practitioners (US); Heffter Research Institute (US); GROF Legacy Training (US); Sage Institute (US); Healing Breakthrough (US); Center for Consciousness Medicine (US)
Individuals Identified as Major Donors, Advisors, or Investors
Individuals are listed solely based on their appearance in publicly available reporting related to funding, advisory, or investment roles, without assessment of personal conduct, intent, or compliance:
Tim Ferriss; Antonio Gracias; Chris Hohn; Kimbal & Christiana Musk; Blake Mycoskie; George Sarlo; Carey Turnbull; Vicky Dulai; Dr. David Bronner
Figure 1. Psychedelic Funders Mind Map (2025), compiled by Psymposia from publicly available tax filings, organizational disclosures, and investigative reporting. Used here for reference and contextual illustration.
Relevant Laws and Regulatory Bodies (U.S.-Focused)
Internal Revenue Service (IRC §§ 501(c)(3), 4940, 4941, 4942, 4943, 4944, 4945)
State Attorneys General (charitable trust enforcement)
Securities and Exchange Commission (disclosure requirements)
Federal Election Commission (PAC and nonprofit coordination rules)
Department of Justice and Federal Trade Commission (antitrust)
Food and Drug Administration (clinical trial and conflict-of-interest rules)
Key Areas of Concern
1. Use of Charitable and Tax-Exempt Assets
Whether nonprofit or university research funded by tax-exempt dollars materially benefits for-profit pharmaceutical or investment entities
Whether charitable funds support media, advocacy, or messaging that advances commercial interests
Whether intellectual property, and / or clinical data has been transferred at fair market value
2. Conflicts of Interest and Governance Overlap
Overlapping boards, donors, officers, and advisors across nonprofits, foundations, venture funds, drug developers, and media entities
Adequacy of disclosure, recusal, and governance safeguards
3. Clinical Trial Integrity
Disclosure of financial interests by investigators
Independence of institutional review boards (IRBs)
Sponsor influence on trial design, outcomes, or publication
4. Market Structure and Access
Whether training, credentialing, or referral organizations function as gatekeepers
Potential anticompetitive effects as psychedelic therapies approach FDA approval
Requested Congressional Action
I respectfully ask Members of Congress and appropriate committees to consider:
Requesting briefings from relevant federal agencies
Evaluating whether existing regulatory frameworks adequately address cross-sector conflicts of interest
Determining whether additional transparency or oversight mechanisms are needed
Conclusion
This request is submitted in the interest of research integrity, charitable accountability, and public trust. Psychedelic therapies may offer meaningful benefits, but public confidence depends on robust safeguards and clear separation between charitable purpose, scientific inquiry, and commercial gain.
I respectfully request that Congress review these issues and determine whether further inquiry is warranted.
Source Materials
INVESTIGATIVE REPORTING; Psymposia (independent media 501(c)3 non-profit) :
https://www.psymposia.com/psychedelic-syndicate-primary-source-documents-silicon-valley-maps-lykos-mdma-fda-billionaires/MEDIA ARTICLE; Psychedelic Alpha (media, incubation, consultancy & advisory):